Мы приняли вашу заявку, свяжемся с вами в ближайшее время.

История
Часть товаров из корзины будет перемещена в статус отложенных и не сможет быть оформлена для заказа, если вы продолжите работу в данном регионе
История

История
История
María Inmaculada García-García, Agustín Sola-Carvajal, Guiomar Sánchez-Carrón, Francisco García-Carmona, Álvaro Sánchez-Ferrer
Department of Biochemistry and Molecular Biology-A, Faculty of Biology, University of Murcia, Campus Espinardo, E-30100 Murcia, Spain
Bioresource Technology 102 (2011) 6186–6191, journal homepage: www.elsevier.com/locate/biortech
Abstract
N-acetyl-D-neuraminic acid aldolase, a key enzyme in the biotechnological production of N-acetyl-D-neuraminic acid (sialic acid) fr om N-acetyl-D-mannosamine and pyruvate, was immobilized as cross-linked enzyme aggregates (CLEAs) by precipitation with 90% ammonium sulfate and crosslinking with 1% FastPrep-CLEAs in biocatalysis, thus reducing production costs and favoring reusability.
© 2011 Elsevier Ltd. All rights reserved.
Article info
Article history:
Received 22 December 2010
Received in revised form 4 February 2011
Accepted 5 February 2011
Available online 3 March 2011
Keywords:
CLEAs
Sialic acid
N-acetylneuraminate lyase
Kinetic parameters
Synthesis
1. Introduction
Although biocatalysts are useful tools for a green and sustainable industry (Sheldon, 2010), their synthetic application is often limited by production and operating costs. Numerous efforts have been devoted to producing robust immobilized catalysts by binding enzyme to a solid carrier, by encapsulation in a organic or inorganic polymeric gel, or by cross-linking of the protein molecules in the form of cross-linked enzyme crystals (CLECs) or cross-linked enzyme aggregates (CLEAs) (Roessl et al., 2010). The latter method not only avoids the inherent dilution of the enzyme in the presence of an inert carrier, but also the need for a highly purified enzyme to start the laborious screening to identify conditions of crystallization that produce catalytic crystals (CLECs). A simple way of preparing CLEAs consists of protein precipitation into an aqueous solution by adding a salt, a water-miscible organic solvent or a polymer (Gupta et al., 2009). In a subsequent step, physical aggregates of the enzyme are cross-linked with a bifunctional agent, the agent of choice being glutaraldehyde, since it is inexpensive and readily available in commercial quantities (Sheldon, 2007). Such cross-linking produces CLEAs, in which the catalytic activity of an individual enzyme is preserved (Sangeetha and Abraham, 2008). Since precipitation is frequently used to purify enzymes, this technology offers the possibility of using semipurified enzymes as starting material, thus reducing costs. In addition, multiple enzyme activities can be simultaneously captured in such aggregates (combi-CLEAs), for cascade or non-cascade conversion (Dalal et al., 2007).
To exploit to its full the simplicity and robustness of CLEAs technology, several key issues need to be resolved, including how to control the particle size of the enzyme aggregates without causing significant diffusion constraints, how to avoid the dramatic modification of some essential e-amino groups by BSA is used, since the internal structure of the aggregates is disturbed, resulting in narrower channels that limit the diffusion of the substrate thought the CLEAs (Cabana et al., 2007).
In this context, special attention should be paid to the two new methods developed to reduce the diffusional limitations of CLEAs. When the substrates of an enzyme are macromolecules, porous cross-linked enzyme aggregates (p-CLEAs) are prepared by adding starch as a pore-making agent, which is latter removed by a-amylase (Wang et al., 2011). In the case of small substrates, our group has described the use of a commercial cell disruptor (FastPrep,), based on a precession movement to recover all the enzyme activity of an acetyl xylan esterase in a few seconds (Montoro-García et al., 2010). This equipment produces a high- speed movement in all directions (vertical and angular motion), which causes cell disruption as a result of the collision between cells and beads in the reaction tube. The effectiveness of the cell disrupting process depends on the rate of the collision and the energy of the impact, which are functions of the speed settings (range, 4.0–6.5 m/s) and the specific gravity of the bead material used. The rate of collision is proportional to the speed, while the energy of impact is proportional to the square of the speed (Müller et al., 1998). In the case of CLEAs, their solid aggregates act as a dispersing material pulverizing each other in a short period of time (30 s) without the need for glass beads.
In order to expand the general use of such FastPrep-CLEAs (FP-CLEAs) in biocatalysis, this methodology was applied to recover the activity of N-acetyl-D-neuraminic acid aldolase (E.C. 4.1.3.3., NAL), a model enzyme that shows all the main characteristic that need to be solved to produce CLEAs. Its activity decreased to 51% after protein crosslinking (0.5% glutaraldehyde lowered the activity to 49% (Mateo et al., 2004). In addition, free NAL is used industrially at basic pHs (wh ere standard CLEAs are not stable) for the condensation of pyruvate and N- acetyl-D-mannosamine (ManNAc) into N-acetyl-D-neuraminic acid (Neu5Ac), an advanced intermediate for obtaining GlaxoSmithK- lime’s antiviral Relenza® (Liese et al., 2006).
2. Methods
2.1. Materials
Pure recombinant Lactobacillus plantarum WFCS1 N-acetyl-D-neuraminic acid aldolase (LpNAL) with the 10-histidine C-terminal extension of pET-52 (Novagen, USA) was produced fr om Escherichia coli BL21(DE3)pLys cells (Novagen, USA) in three simple purification steps, as previously reported (Sánchez-Carrón et al., 2011). Sugars were from Nacalai Tesque (Kyoto, Japan). High purity ammonium sulfate (BioUltra) were obtained from Fluka (Madrid, Spain). Other reagents were from Sigma (Madrid, Spain).
2.2. CLEAs production
Aggregation tests with ammonium sulfate and the following steps were carried out at 4 °C by adding to 1 mL of a 10 mg/mL pure LpNAL in 20 mM sodium phosphate buffer pH 7.5, the required volume of saturated ammonium sulfate solution to bring the mixture to the desired degree of saturation with the salt. Pilot aggregation tests with acetonitrile or with tert-butanol (final concentrations up to 90% v/v) were carried out in the same way, except that cold solvent was added. After 1 h, the appropriate amount of a freshly prepared 25% (v/v) aqueous solution of centrifuged (16,000g x 2 min), after which the enzyme activity in the supernatant and in precipitates was determined.
Test assays yielded active aggregates when using ammonium sulfate at 90% saturation and a 4-h cross-linking period with 1% centrifugation.
AS-CLEAs.
Finally, SB-CLEAs. All the preparations of the different CLEAs described above were stable for several weeks when stored at 4 °C.
2.3. Enzyme assays
For the standard assay of the synthetic activity of N-acetyl-D-neuraminic acid aldolase, i.e., obtaining Neu5Ac from pyruvate and ManNAc, the enzyme (generally, 0.1 mg/mL of CLEAs) was incubated at 37 °C with the above substrates at the indicated concentration of 100 mM ManNAc and 30 mM Pyr in 20 mM sodium phosphate pH 7.5. The reaction was stopped by centrifuging out the CLEAs (16,000g x 2 min). The sample from the supernatant were subjected to normal-phase HPLC (LC-20A, Shimazu) using an AMINO-UK column (Imtakt Corporation, Kyoto, Japan), a mobile phase consisting of 58% acetonitrile–42% 50 mM ammonium acetate and a flow-rate 0.4 mL/min at 60 °C. The elution profile was monitored both at 210 nm and with an ELSD-II detector (Shimadzu, Japan). In these conditions, the retention time (Rt) for Neu5Ac and ManNAc were 10.34 and 4.22 min, respectively. One unit of activity was defined as the amount of enzyme required to synthesize 1 lmol of Neu5Ac per minute under the above conditions (Sánchez-Carrón et al., 2011). Enzymatic activity was obtained from three repeated experiments.
2.4. Scanning electron microscopy of CLEAs
Droplets from CLEAs suspensions were placed on moist filter papers and dehydrated serially (10 min, each step) by passage through a graded series of acetone solutions (30%, 50%, 70%, 90% and 100%) and dried at the critical point of CO₂ (Montoro-García et al., 2010). After gold sputtering, the samples were visualized with a Jeol T6.100 (Japan) scanning electron microscope (SEM) operated at 15 kV.
2.5. CLEA dispersion and sizing of the resulting particles
Two methods were used for the disruption of the aggregates. One approach, which is widely used, consisted of vortexing at medium speed with a Heidolph (Germany) Reax Control vortex. In the other (novel) approach, the CLEAs were subjected to reciprocating mixing using a FastPrep-24 sample preparation system (M.P. Biomedicals), CA, USA) at a setting of 6.0 m/s (Montoro-García et al., 2010). The degree of dispersion attained by both procedures was monitored by bright-field optical microscopy at a final enlargement of 40x, taking digital images that were analyzed planimetrically by image processing (MIP 4.5 image analysis software, Digital Image System, Barcelona, Spain).
2.6. Enzyme stability assay
The pH-stability was monitored by incubating the enzyme (either in soluble or in CLEA form) in a solution of 20 mM glycine buffer pH 9.0 at 50 °C, taking samples after the periods of time specified to determine enzyme activity in the standard assay at 37 °C, using ManNAc and pyruvate as substrates. In a similar way, the thermal stability was recorded by incubating the enzyme (in a free and aggregates form) in a solution of 20 mM glycine buffer pH 9.0 at the indicated temperatures (40–80 °C). After cooling, enzyme activity was determined in standard assay at 37 °C.
2.7. Computer analysis
Lysines and the Grand average hydropathicity index (GRAVY) of LpNAL and BSA were calculated using the ProtParam tool from Expasy Proteomic server ( http://www.expasy.org/tools/protpa- ram.html). Negative GRAVY values indicate hydrophilic proteins (Kyte and Doolittle, 1982).
3. Results and discussion
3.1. Optimization of CLEAs production
Pure L. plantarum N-acetyl-D-neuraminic acid aldolase (LpNAL) was used for preparation of the aggregates, since this enzyme has great potential as a biocatalyst due to its high stability at alkaline pHs, its high over-expression in E. coli and its simple purification procedure (Sánchez-Carrón et al., 2011). In a preliminary screen to select a suitable agent for protein aggregation, three different substances were tested up to 90% concentration, either to change the hydration state of the enzyme molecules (ammonium sulfate) or to alter the electrostatic constant of the solution (acetone and acetonitrile). After 1 h, the appropriate amount of vortexing (Table 1), whereas organic solvents eliminated the activity, pointing to a solvent-triggered denaturing effect.
Similar findings have been reported for other CLEAs (Gupta et al., 2009; Hara et al., 2008; Montoro-García et al., 2010), wh ere ammonium sulfate was also the selected precipitant.
In order to improve not only this low amount of recovered activity but also the stability of the above AS-CLEAs, BSA as a proteic feeder (Shah et al., 2006) (see below, Section 3.2).
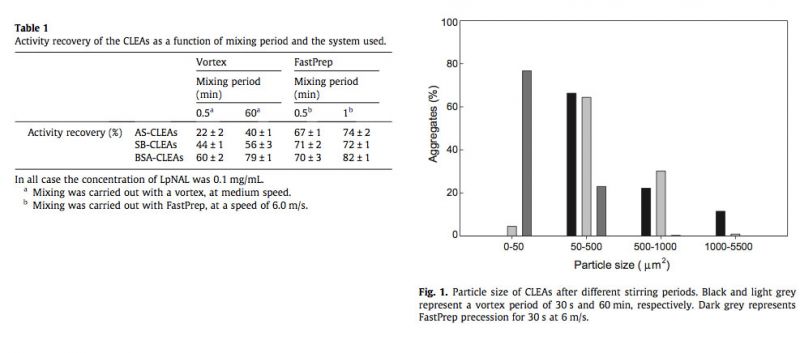
Planimetric analysis of the different aggregates also showed the influence of the time and dispersing device used (Supplementary Fig. S1). All mechanical treatments fragmented the CLEAs into particles, but most of the aggregates after 30-s vortexing (Fig. 1, black bars), were distributed in particle sizes up to 1000 μm², while 11% were larges (reaching 5000 μm²). After 1-h vortexing (Fig. 1, light grey bars), 60% of particles were in the 50–500 μm² range, whereas after 60-s of FastPrep treatment (Fig. 1, dark grey bars), 86% of particles were in the smallest range (0–50 μm²).
The last treatment, which is also the one that yielded the maximal recovery of the activity, clearly does not disturb the individual CLEAs, and, hence, the enzyme structure (as is evident from the SEM analysis, see below), preventing that particle from being compressed close together with concomitant mass-transport limitations. Similar results with FastPrep were also reported for acetyl xylan esterase CLEAs, although the particle size obtained was slightly greater, in the 0–149 μm² range (Montoro-García et al., 2010). Thus, it is clear from Table 1 and Fig. 1 that the FastPrep system could be of general application to produce efficient CLEAs, regardless of the protocol used to prepare them, and only CLEAs produced with this system were used in the rest of the present work.
3.2. Structure of FastPrep-CLEAs
Scanning electron microscopy (SEM) of the FastPrep-CLEAs produced in this paper revealed amorphous structures which differed depending on the protocol used. Thus, the CLEAs obtained with ammonium sulfate (AS-CLEAs) formed less structured and more coarse-grained aggregates (Supplementary Fig. S1a) than those formed by ammonium sulfate and BSA, which both have a clear negative Grand average of hydropathicity (GRAVY) number (-0.301 and -0.429, respectively).
3.3. Kinetic characterization of FastPrep-CLEAs
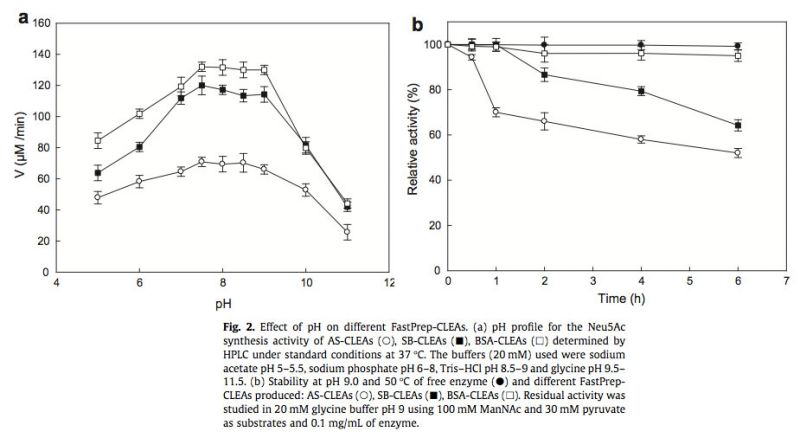
The catalytic activity of the three different FastPrep-CLEAs produced in this paper was investigated over a wide pH range (Fig. 2a). All the CLEAs exhibited a broad bell-shape pH dependence of the catalytic activity, similar to that seen for free enzyme (Sánchez-Carrón et al., 2011), but with a pH shift towards alkaline, with an optimum of pH 7.5 in contrast to the pH 7.0 of free enzyme. This shift could have resulted fr om the change in acidic and basic amino acid side chain ionization in the microenvironment around the active site, which was produced by the freshly produced interaction between basic residues of the enzyme and BSA (Fig. 2a, open squares) and sodium borohydride (Fig. 2a, filled squares) compared with ammonium sulfate alone (Fig. 2a, open circles) was more evident at alkaline pHs, giving rise to a fully active catalyst at pH 9.0, which is one of the demands for the chemoenzymatic synthesis of Neu5Ac from the inexpensive N-acetyl-D-glucosamine (Liese et al., 2006).
In addition, the biocatalyst has to be stable at such alkaline pH values, as is seen from Fig. 2b, wh ere it is clear that only BSA-CLEAS due to cross-linking were observed (data not shown), since NALs are very thermostable proteins with melting points (Tm) of about 74 °C at pH 9.0 (Sánchez-Carrón et al., 2011) and an optimal temperature about of 70–80 °C (Aisaka et al., 1991).
The kinetic parameters for the synthetic activity were also investigated with the best FastPrep-CLEAs obtained with glutaraldehyde. The latter reagent was also responsible for the decrease in the turnover and in the catalytic efficiency (kcat/KM) of the enzyme for both substrates (Table 2), clearly indicating an attack on the catalytic lysine of NAL. Nevertheless, and in spite of the values shown in Table 2, these CLEAs are of biotechnological relevance. The apparent KM for ManNAc is lower than that reported for the enzymatic synthesis of Neu5Ac, with a KM values of 402 mM (Kragl et al., 1991), which needs concentrations about 0.7 M to reach maximum activity. On the other hand, the lower KM of pyruvate is also advantageous, since a high pyruvate concentration in the solution after the bioconversion hinders product recovery, due to the fact that pyruvate and Neu5Ac exhibit similar pKa values in the region of 2.2 (Kragl et al., 1991), making a tedious ion-change chromatography step necessary (Sugai et al., 1995).
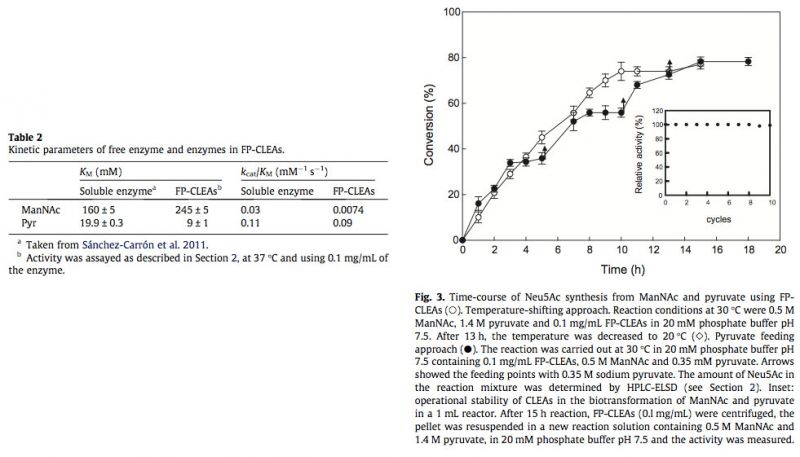 3.4. Potential production of Neu5Ac
3.4. Potential production of Neu5Ac
To investigate the potential of LpNAL CLEAs for the production of the Neu5Ac in aqueous medium, the condensation of ManNAc and pyruvate by FP-CLEAs was studied. In this enzymatic reaction, an excess of pyruvate over ManNAc is generally used to achieve high yield of Neu5Ac, because the equilibrium tends towards pyruvate and ManNAc (Keq, 28.7 M-1 at 25 °C) (Kragl et al., 1991). For this reason, the FP-CLEAs reaction was carried out at 30 °C with an almost threefold molar excess of pyruvate over ManNAc (1.4 M/0.5 M) to archive a high reaction rate. When the reaction was approaching its equilibrium point, after about 13 h (Fig. 3, open circles), the reaction temperature was reduced to 20°C (Fig. 3, open diamonds) to obtain 77% conversion. Fig. 3 also shows the comparison of the temperature shifting reaction with another reaction in which pyruvate (0.35 M) was fed three times after the equilibrium point was achieved at 5, 10 and 13 h (Fig. 3, arrows), respectively. In both cases, the same yield was obtained, indicating the advantage of the temperature-shifting approach due to its simplicity and no need for repetitive additions of pyruvate. In addition, the yield obtained with FP-CLEAs from ManNAc was higher than that obtained with commercial Neu5Ac aldolase (60%) (Sugai et al., 1995), but lower than that obtained with a 10-fold higher concentration of enzyme in a free form (Blayer et al., 1996), which clearly avoids the possibility of enzyme inactivation produced during longer periods of reaction, as a result of pyruvate degradation (Blayer et al., 1996).
Having demonstrated Neu5Ac production with LpNAL FP-CLEAs, their stability during multiple reuses was investigated, using the temperature-shifting approach. Typically, reactions (1 mL) reached completion within 15 h, and at least 10 cycles were possible without any significant loss of activity (Fig. 3, inset). Such stability is the same as that observed when Neu5Ac aldolase is used in an industrial process, with a known solid support like Eupergit-C (Mahmoudian et al., 1997) or when it is immobilized into cross-linked inclusion bodies (CLIBs) (Nahàlka et al., 2008). This strongly supports the value of using hardened CLEAs in combination with the FastPrep system for dispersion, to yield FP-CLEAs with excellent resistance to mechanical stress, allowing high recoveries of catalyst after many cycles of use. These ten cycles without loss of activity in CLEAs has also been described for lipase (Gupta et al., 2009), R-oxynitrilase (van Langen et al., 2005) and acetyl xylan esterase (Montoro-García et al., 2010), but contrast with the four reuses possible with hydroxynitrile lyase (Cabirol et al., 2008) or subtilisin (Sangeetha and Abraham, 2008).
4. Conclusions
The results presented here highlight the potential of using hardened CLEAs (FP-CLEAs in the sequential enzymatic reaction for producing Neu5Ac from inexpensive N-acetyl-D-glucosamine, catalyzed by N-acetyl-D-glucosaminine-2-epimerase and N-acetyl-D-neuraminic acid aldolase.
Acknowledgements
This study was partially supported by Spanish grants from MEC-FEDER (BIO2007-62510) and from Programa de Ayuda a Grupos de Excelencia de la Región de Murcia, Fundación Séneca (04541/GERM/06, Plan Regional de Ciencia y Tecnología 2007– 2010). M.I.G.G. and A.S.C. are holders of predoctoral research grants associated to the above project from Fundación Séneca. G.S.C. is a holder of a predoctoral research grant (FPU) from Ministerio de Educación y Ciencia, Spain. We are very grateful to Sofía Jiménez García, a Research Support Staff member from the University of Murcia, for her technical support, and also to María García, María Teresa Castels and Fara Saez from the Servicio de Ayuda a las Cien- cias Experimentales (SACE, Murcia, Spain) for their help in SEM images.
Appendix A. Supplementary data
Supplementary data associated with this article can be found, in the online version, at doi:10.1016/j.biortech.2011.02.026.
References
Aisaka, K., Igarashi, A., Yamaguchi, K., Uwajima, T., 1991. Purification, crystallization and characterization of N-acetylneuraminate lyase from Escherichia coli. Biochem. J. 276, 541–546.
Aytar, B.S., Bakir, U., 2008. Preparation of cross-linked tyrosinase aggregates. Process. Biochem. 43, 125–131.
Blayer, S., Woodley, J.M., Lilly, M.D., 1996. Characterization of the chemoenzymatic synthesis of N-acetyl-D-neuraminic acid (Neu5Ac). Biotechnol. Prog. 12, 758– 763.
Cabana, H., Jones, J.P., Agathos, S.N., 2007. Preparation and characterization of cross- linked laccase aggregates and their application to the elimination of endocrine disrupting chemicals. J. Biotechnol. 132, 23–31.
Cabirol, F.L., Tan, P.L., Tay, B., Cheng, S., Hanefekd, U., Sheldon, R.A., 2008. Linum usitatissium hydroxynitrile lyse cross-linked enzyme aggregates: a recyclable enantioselective catalyst. Adv. Synth. Catal. 350, 2329–2338.
Cao, L., van Rantwijk, F., Sheldon, R.A., 2000. Cross-linked enzyme aggregates: a simple and effective method for the immobilization of penicillin acylase. Org. Lett. 2, 1361–1364.
Dalal, S., Kapoor, M., Grupta, M.N., 2007. Preparation and characterization of combi- CLEAs catalyzing multiple non-cascade reactions. J. Mol. Catal. B: Enzyme 44, 128–132.
Dong, T., Zhao, L., Huang, Y., Tan, X., 2010. Preparation of cross-linked aggregates of aminoacylase from Aspergillus melleus by using bovine serum albumin as an inert additive. Bioresour. Technol. 101, 6569–6571.
Gupta, P., Dutt, K., Misra, S., Raghuwanshi, S., Saxena, R.K., 2009. Characterization of cross-linked immobilized lipase from thermophilic mould Thermomyces lanuginosa using glutaraldehyde. Bioresour. Technol. 100, 4074–4076.
Hara, P., Hanefeld, U., Kanerva, L.T., 2008. Sol–gels and cross-linked aggregates of lipase PS from Burkholderia cepacia and their application in dry organic solvents. J. Mol. Catal. B: Enzyme 50, 80–86.
Kragl, U., Gygax, D., Ghisalba, O., Wandrey, C., 1991. Enzymatic two-step synthesis of N-acetyl-neuraminic acid in the enzyme membrane reactor. Angew. Chem. Int. Ed. Engl. 30, 827–828.
Kyte, J., Doolittle, R.F., 1982. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132.
Liese, A., Seelbach, K., Buchholz, A., Haberland, J., 2006. Processes. In: Liese, A., Seelbach, K., Wandrey, C. (Eds.), Industrial Biotransformations. Wiley-VCH, Weinheim, Germany, pp. 457–460.
Mahmoudian, M., Noble, D., Drake, C.S., Middleton, R.F., Montgomery, D.S., Piercey, J.E., Ramlakhan, D., Todd, M., Dawson, M.J., 1997. An efficient process for production of N-acetylneuraminic acid using N-acetylneuraminic acid aldolase. Enzyme Microbiol. Technol. 20, 393–400.
Mateo, C., Palomo, J.M., van Langen, L.M., van Rantwijk, F., Sheldon, R.A., 2004. A new, mild crosslinking methodology to prepare crosslinked enzyme aggregates. Biotechnol. Bioeng. 86, 273–276.
Matijosyte, I., Arends, I.W.C.E., de Vries, S., Sheldon, R.A., 2010. Preparation and use of cross-linked enzyme aggregates (CLEAs) of laccases. J. Mol. Catal. B: Enzyme 62, 142–148.
Montoro-García, S., Gil-Ortiz, F., Navarro-Fernández, J., Rubio, V., García-Carmona, F., Sánchez-Ferrer, A., 2010. Improved cross-linked enzyme aggregates for the production of desacetyl b-lactam antibiotics intermediates. Bioresour. Technol. 101, 331–336.
Müller, F.M., Werner, K.E., Kasai, M., Francesconi, A., Chanock, S.J., Walsh, T.J., 1998. Rapid extraction of genomic DNA from medically important yeasts and filamentous fungi by high-speed cell disruption. J. Clin. Microbiol. 6, 1625– 1629.
Nahàlka, J., Vikartovská, A., Hrabárová, E., 2008. A crosslinked inclusion body process for sialic acid synthesis. J. Biotechnol. 134, 146–153.
Roessl, U., Nahálka, J., Nidetzky, B., 2010. Carrier-free immobilized enzymes for biocatalysis. Biotechnol. Lett. 32, 341–350.
Sánchez-Carrón, G., García-García, M.I., López-Rodríguez, A.B., Jiménez-García, S., Sola-Carvajal, A., García-Carmona, F., Sánchez-Ferrer, A., 2011. Molecular characterization of a novel N-acetylneuraminate lyase from Lactobacillus plantarum WCFS1. Appl. Envirom. Microbiol, doi:10.1128/AEM.02927-10 [Epub ahead of print].
Sangeetha, K., Abraham, T.E., 2008. Preparation and characterization of cross-linked enzyme aggregates (CLEAs) of subtilisin for controlled release applications. Int. J. Biol. Macromol. 43, 314–319.
Schoevaart, R., Wolbers, M.W., Golubovic, M., Ottens, M., Kieboom, A.P.G., van Rantwijk, F., van der Wielen, L.A.M., Sheldon, R.A., 2004. Preparation, optimization, and structures of cross-linked enzyme aggregates (CLEAs). Biotechnol. Bioeng. 87, 754–762.
Shah, S., Sharma, A., Gupta, M.N., 2006. Preparation of cross-linked enzyme aggregates by using bovine serum albumin as a proteic feeder. Anal. Biochem. 351, 207–213.
Sheldon, R.A., 2007. Cross-linked enzyme aggregates (CLEAÒs): stable and recyclable biocatalysts. Biochem. Soc. Trans. 35, 1583–1587.
Sheldon, R.A., 2010. Cross-linked enzyme aggregates as industrial biocatalysts. In: Shioiri, T., Izawa, K., Konoike, T. (Eds.), Pharmaceutical Process Chemistry. Wiley-VCH, Weinheim, Germany. doi:10.1002/9783527633678.ch8.
Sugai, T., Kuboki, A., Hiramatsu, S., Okazaki, H., Ohta, H., 1995. Improved enzymatic procedure for a preparative-scale synthesis of sialic acid and KDN. Bull. Chem. Soc. Jpn. 68, 3581–3589.
Tyagi, R., Batra, R., Grupta, M.N., 1999. Amorphous enzyme aggregates: stability towards heat and aqueous-organic cosolvents mixtures. Enzyme Microbiol. Technol. 24, 348–353.
van Langen, L.M., Selassa, R.P., van Rantwijk, F., Sheldon, R.A., 2005. Cross-linked aggregates of (R)-oxynitrilase: a stable, recyclable biocatalyst for enantioselective hydrocyanation. Org. Lett. 7, 327–329.
Wang, M., Qi, W., Yu, Q., Su, R., He, Z., 2010. Cross-linking enzyme aggregates in the macropores of silica gel: a practical and efficient method for enzyme stabilization. Biochem. Eng. J. 54, 168–174.
Wang, M., Jia, C., Qi, W., Yu, Q., Peng, X., Su, R., He, Z., 2011. Porous-CLEAs of papain: application to enzymatic hydrolysis of macromolecules. Bioresour. Technol. 102, 3541–3545.
Wilson, L., Betancor, L., Fernandez-Lorente, G., Fuentes, M., Hidalgo, A., Guisan, J.M., Pessela, B.C.C., Fernandez-Lafuente, R., 2004. Crosslinked aggregates of multimeric enzymes: a simple and efficient methodology to stabilize their quaternary structure. Biomacromolecules 5, 814–817.
Abbreviations: CLEAs, cross-linked enzyme aggregates; AS, ammonium sulfate; FastPrep-CLEAs; Neu5Ac, N-acetylneuraminic acid (sialic acid); NAL, N-acetylneuraminate lyase; ManNAc, N-acetyl-D-mannosamine; Pyr, pyruvate; LpNAL, Lactobacillus plantarum NAL; 7-ACA, 7-aminocephalosporanic acid.
* Corresponding author. Tel.: +34 868884770; fax: +34 868884147. E-mail address: alvaro@um.es (Á. Sánchez-Ferrer).
С помощью личного кабинета Вы сможете:
Сравнение